Humic Acids and Phosphorus
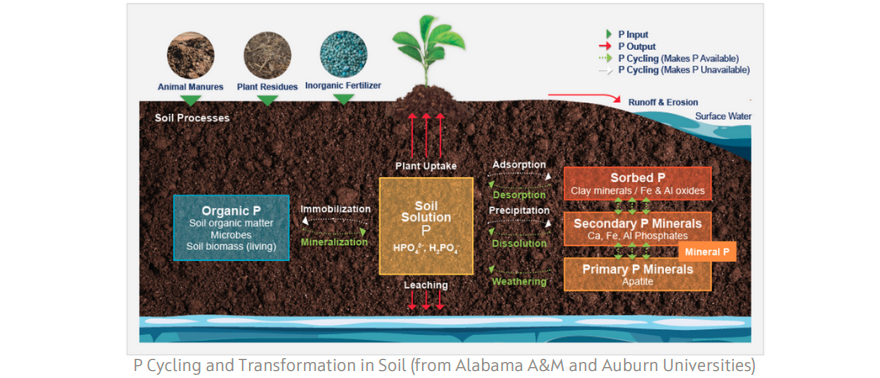
Soil phosphorus is found in two forms, namely organic and inorganic (Mineral). These two forms together make up the total of the soil’s phosphorus. Although total soil phosphorus is generally high, with concentrations ranging from 200 to 6,00 pounds per acre, up to 90 percent of this phosphorus is immobile and unavailable to the plant. Approximately 30 to 65 percent of total soil phosphorus is in organic forms, which are not plant-available, while the remaining 35 to 70 percent is in inorganic forms.
Organic phosphorus: this fraction includes plant/animal residues and soil microorganisms. Microorganisms play a crucial role in processing and transforming this organic phosphorus into plant-available forms. The presence of humic acids significantly promotes microbe growth and facilitates the mineralization process.
Sorbed phosphorus: This phosphorus pool is comprised of inorganic phosphorus attached to clay surfaces, iron (Fe), aluminum (Al), and calcium (Ca) oxides in soil. The phosphorus in this pool is released slowly for plant uptake. Humic molecules can form complexes with phosphate ions, keeping P available to plants and less adsorbed to clay surfaces.
Mineral phosphorus: This phosphorus pool comprises the soil’s primary and secondary phosphate minerals. Examples of primary phosphorus minerals include apatite, strengite, and variscite. The secondary phosphorus minerals include calcium (Ca), iron (Fe), and aluminum (Al) phosphates. The release of phosphorus from this pool is prolonged and occurs when the mineral weathers and dissolves in soil water. Humic acids promote Mycorrhizal Fungi that feed on organic carbons in your soil They produce a substance called Glomalin that helps break down Tricalcium Phosphate and many other minerals.