Humic Acids and Potassium
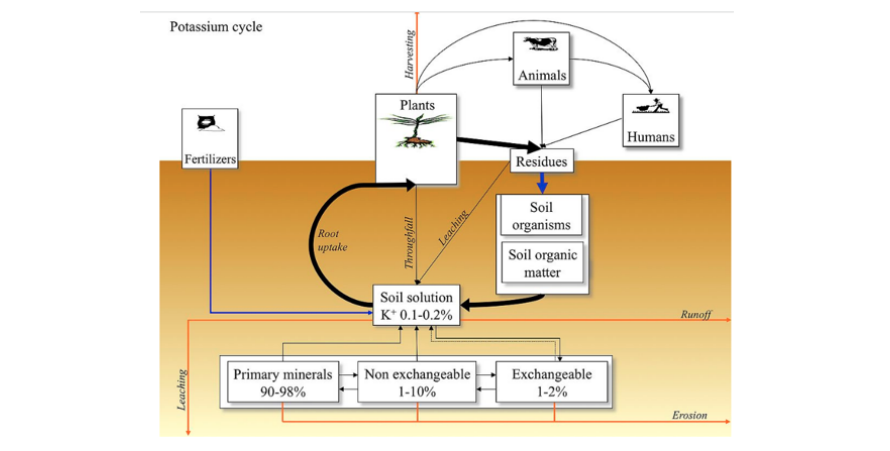
Potassium in Soil Solution
Potassium cations (K+) dissolved in the soil solution account for 0.1-0.2% of total soil potassium. Plant roots take up K as the K+ ion from the soil solution. Potassium is taken up by plants in large quantities
Exchangeable Potassium
Exchangeable K includes those K+ ions adsorbed (by electrostatic forces) and released on clay and organic colloids. The exchangeable K accounts for 1-2% of total soil K. Humic substances has negative charges with carbon functional groups so they have has CEC values. Application of humic acids would greatly improve CEC of soil media so that more K ions can be adsorbed on CEC sites and less prone to leaching loss.
Non-exchangeable Potassium
Non-exchangeable K refers to K+ ions adsorbed in the interlayer spacing of clay minerals, such as illite, vermiculite, and chlorite. This form of K accounts for 1-10% of total soil potassium. Release of potassium fixed by expanding silicate clays is considered of practical importance in soil fertility. Humic and fulvic acids are expected to play a definite role in liberating this fixed K, because of their chelating power. In terms of percentage of the total K fixed, 9 to 28% can be released by humic acids. The percentages K released by humic and fulvic acids were similar from both montmorillonite and illite, but based on absolute values, humic and fulvic acids extracted less K (mg/100 g) from illite than montmorillonite.